Congratulations to Dr. Jin Zhang on Publishing a Research Article in Angewandte Chemie International Edition
Energy Efficiency Limit in CO-to-Ethylene Electroreduction and the Method to Advance Toward
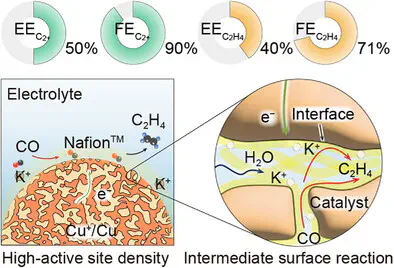
The electrified synthesis of high-demand feedstocks (C2H4) from CO and H2O through a CO electroreduction (COR) protocol is attractive for large-scale applications; however, a high reaction potential and modest Faradaic efficiencies (FEs) limit its practical energy efficiency (EE). In this study, a quantitative reaction–transport model was constructed to analyze the root causes of low performance in COR, which revealed low volumetric exchange current density and limited intermediate surface reaction as key factors, constraining CO-to-C2+ and CO-to-C2H4 conversion energetics and selectivities. Consequently, a robust, high active-site density electrode, featuring nanometer-scale interspacing between the active, Nafion-wrapped Cu+–Cu nanosheet catalysts, was designed. This design increases volumetric COR activity with an efficient intermediate surface reaction mechanism for C2H4 production, substantially lowering the full-cell COR potential to 1.87 V at 4 A in a 25 cm2 membrane electrode assembly, thereby achieving a record >50% C2+ EE with a 90 ± 1% FE along with a >40% C2H4 EE with a 71 ± 1% FE throughout stable >100 h operation. Similarly designed high-volumetric-activity Bi and Ag nanosheet catalysts enabled >60% and >55% EEs for the CO2-to-formate and CO2-to-CO electroreduction, demonstrating the broader applicability of our electrochemical activity and EE enhancement concept on a three-phase interface.
The related research findings were published in Angewandte Chemie International Edition under the title "Energy Efficiency Limit in CO-to-Ethylene Electroreduction and the Method to Advance Toward".
Article link: https://onlinelibrary.wiley.com/doi/full/10.1002/anie.202502690