Congratulations to Dr. Yongcheng Xiao on Publishing a Research Article in Journal of Environmental Sciences
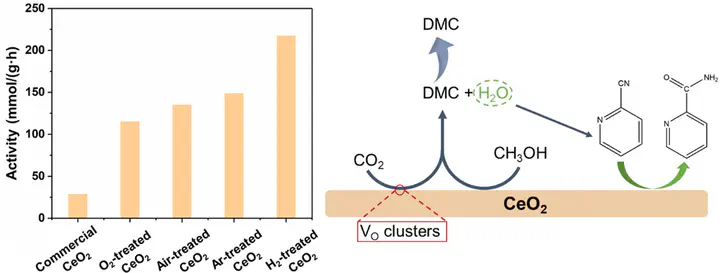
Boosting dimethyl carbonate synthesis from CO2 and methanol through oxygen vacancy engineering on CeO2 under thermodynamically favorable conditions
The direct conversion of greenhouse gas CO2 and low-cost CH3/sub>OH into valuable dimethyl carbonate (DMC) offers a promising low-carbon synthetic pathway, but the slow CO2 activation kinetics and entropy-decreasing nature of this reaction significantly restrict DMC yield to below 1 %. In this work, 2-cyanopyridine (2-CP) was employed as a dehydrating agent to suppress the reverse reaction between DMC and H2O, shifting the thermodynamic equilibrium in favor of DMC production. Under this thermodynamic unconstrained condition, increasing oxygen vacancies, especially in the form of oxygen vacancy clusters, promotes catalytic activity significantly. We achieve a catalytic activity of 211 mmol/(g·h) at 140 °C on H2-treated, oxygen-vacancy-clusters-rich CeO2 in the presence of 2-CP, a 1.6-fold increase compared to the activity with air-treated CeO2 under identical conditions. The DMC yield reaches 8.54 % in a 20 mL CH3OH solution with 2-CP, surpassing the calculated DMC yield of about 0.66 % from the reaction equilibrium constant under the same conditions and without using the dehydrating agent. This work suggests the importance of using a dehydrating agent and also highlights oxygen vacancy clusters as pivotal active sites to promote DMC synthesis. Achieving sustainable DMC synthesis requires further exploration, encompassing strategies such as methods for regeneration of 2-CP.
The related research findings were published in Journal of Environmental Sciences under the title "Boosting dimethyl carbonate synthesis from CO2 and methanol through oxygen vacancy engineering on CeO2 under thermodynamically favorable conditions".
Article link: https://www.sciencedirect.com/science/article/pii/S1001074224002870